Article #3 of a 7 Part Series
Neurons of the brain are ‘electrical’ cells, but their electricity is not the same as that flowing in the wires in your home. Brain’s form of electrical current is produced by movement of charged atoms across a neuron’s cell membrane.
The method by which electrical current of neurons becomes a very fast messaging system is probably the most difficult concept for people new to science to get their mind around. So today we will not be discussing signaling within the brain but will rather focus on the environment necessary for the system to work.
A consistently precise chemical composition of the fluid surrounding neurons is essential for brain’s charged atoms, called ions, to produce neuron electrical current. Thus, brain’s quality-control system is like no other fluid-control system in the body.
Brain Electricity
Brain electricity depends upon the geometry of the fluid filled space around neurons modeled by engineers is described as a network of pores and tunnels less than 100 nanometers across. A nanometer is one part of a meter that is divided into a billion parts. These spaces are sufficiently small that the process of chemical diffusion of ions is a highly effective mechanism for communication between cells.
Diffusion is when molecules relocate by moving through a solution from the place where their concentration is high to another place where their concentration is low.
Ions diffuse in the watery fluids of the body because they carry a charge that interacts with water molecules. The quality and quantity of an ion’s charge is governed by the atom’s lack of a match between its number of protons, positive particles and electrons, negative particles.
When the concentration of an ion is not the same inside and outside a cell like a neuron, the ion can use any open channel in the cell’s membrane to move toward the fluid compartment where its concentration is lower. For diffusion of ions to be channeled into a coordinated flow of electricity in the brain, it is critical that the ionic composition of brain fluid outside neurons be as tightly regulated as ionic composition of the fluid inside the cell.
Brain Fluid
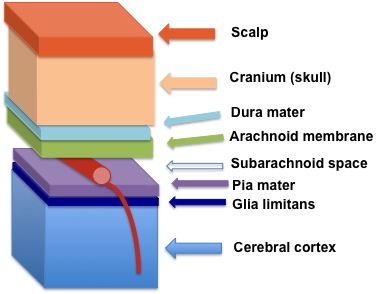
Meninges
The meninges are a series of membranes covering and penetrating the brain as in the diagram below. An ultra-thin membrane named pia mater surrounds brains arteries. The name pia mater comes from Medieval Latin and translates to tender mother. The space created by the presence of pia mater around arterial vessels is known as the Virchow-Robin space, named after the two investigators who discovered its presence.
The Virchow-Robin space ends where brain capillaries begin. There it is eliminated by fusion of the capillary cells with the membranes of brain cells called astrocytes, star-shaped cells.
Virchow-Robin space shields the fluid around neurons from all proteins and other large molecules leaked from arterial blood vessels. It also clears waste-containing fluid from around neurons created by metabolic cellular activity. Waste containing fluid flows through the pia mater into the Virchow-Robin space and then drains into the lymphatic system of the head for return to the heart.
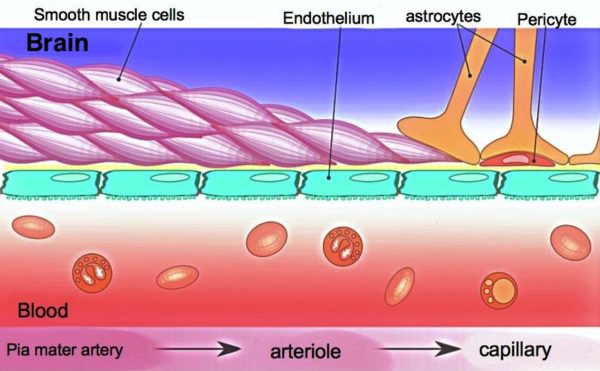
Blood Brain Barrier
About 98% of blood’s small molecules do not enter the brain through its capillary system because of the blood brain barrier. Brain capillaries are also not permeable to water molecules. Elsewhere in the body, direct exchange of water and small water-soluble molecules between blood and fluid surrounding cells is unconstrained at capillaries.
The blood brain barrier is created by tight lacing together of capillary cells by protein formations called adherens junctions. The junctions assure that all molecules entering the brain from the capillaries must go through, rather than around capillary cells. Within capillary cells there are multiple mechanisms for regulation of the passage of molecules to the brain.
An additional layer of physical and operational support for the blood brain barrier comes from membrane projections of another brain cell named astrocyte. The astrocytes’ foot-like structures make direct contact with brain capillaries to provide mechanical support. Astrocytes also secrete chemical factors to block entry to the brain of peripheral immune system cells.
Cerebrospinal Fluid
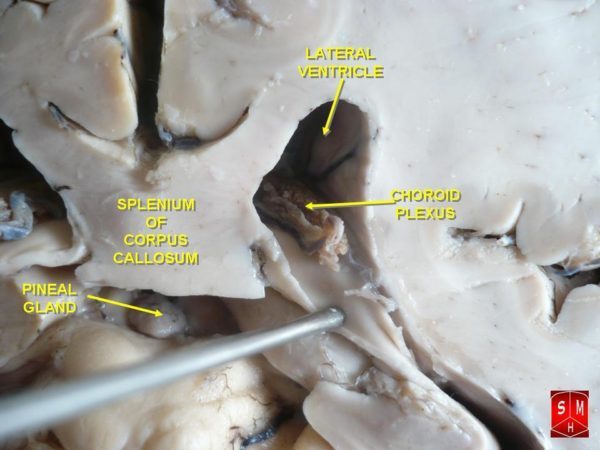
Cerebrospinal fluid circulation is exclusive to the brain. Cerebrospinal fluid is secreted by the ependymal cells lining the brain’s inner chambers, the ventricles. It travels through the chambers and around the spinal cord to the top of the brain where it enters the lymphatics for return to the heart.
Each of the brain’s inner chambers, the ventricles, possesses a structure called the choroid plexus. The choroid plexus provides the brain access to water, selected small molecules, growth factors and nutrients from blood.
The interior of a choroid plexus is a convoluted vascular network of loose connective tissue and large capillaries. Most blood borne small molecules pass around these capillary cells.
Fluid within the choroid plexus must be processed through its outer layer of ependymal cells to become cerebrospinal fluid. Water molecule transfer through the ependymal cells of the choroid plexus is managed separately from transfer of nutrients and growth factors.
Ependymal cells also manufacture and secrete a wide variety of molecules to promote brain health. Additionally, the choroid plexus is active in the clearance from the brain of drugs and pollutants.
In humans about 500 milliliters of cerebrospinal fluid is made per day. The volume of the entire system is only about 150-200 milliliters. Therefore, cerebrospinal fluid is replaced about 2-4 times a day.
Cerebral Blood Flow
Although the brain restricts entry of many substances from blood, a steady cerebral blood flow to deliver glucose, oxygen and growth factors is critical for brain tissue survival.
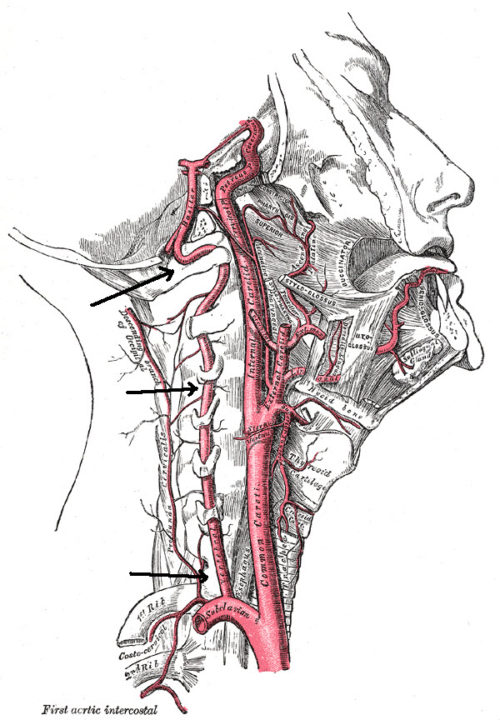
Two major sets of arteries on the right and left side of the body provide blood that is rich in oxygen and glucose to the brain. They are the internal branch of the carotid arteries of the neck and the vertebral arteries, pointed out by the arrows in the illustration.
The internal carotid arteries supply blood to the front and middle portions of the brain. The vertebral arteries supply the back of the brain.
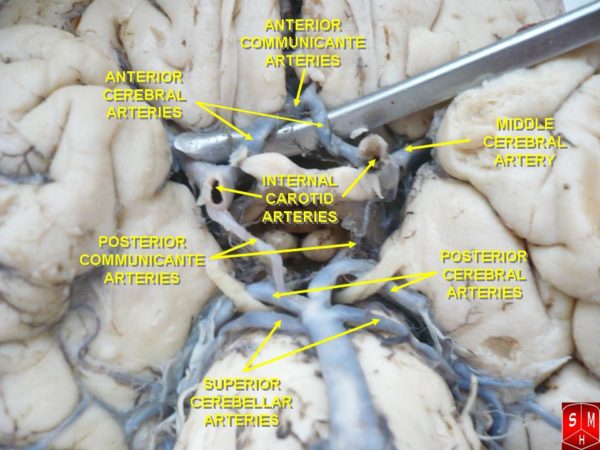
The brain’s arteries spread over its surface before they penetrate deep brain tissue. The anterior carotid and posterior vertebral circulation connect to each other by the posterior communicating arteries at the Circle of Willis, which lies at the base of the brain.
This arrangement serves as a safety net. If one part of the cerebral circulation becomes injured or blocked, blood flow from the other vessels can be shunted through the Circle of Willis to preserve perfusion of most of the brain.
Veins returning from brain capillary beds are like those found elsewhere in the body. When veins draining the anterior brain reach the surface, they empty into large sinuses in the dura mater of the membranes covering the brain. These sinuses come together in the posterior brain and empty into the internal jugular veins to the heart.
Venus return from the posterior surface of the brain and from the cervical spinal cord travels to the heart by way of the vertebral arteries and large veins of the chest.
While cerebral blood flow is critical for providing nutrients and oxygen, the brain must be choosy about how much contact it has with blood in order to maintain its electrical currents. Leakage of blood directly onto brain tissue is toxic to neurons.
Margaret Thompson Reece PhD, physiologist, former Senior Scientist and Laboratory Director at academic medical centers in California, New York and Massachusetts is now Manager at Reece Biomedical Consulting LLC.
She taught physiology for over 30 years to undergraduate and graduate students, at two- and four-year colleges, in the classroom and in the research laboratory. Her books “Physiology: Custom-Designed Chemistry”, “Inside the Closed World of the Brain”, and her online course “30-Day Challenge: Craft Your Plan for Learning Physiology”, and “Busy Student’s Anatomy & Physiology Study Journal” are created for those planning a career in healthcare. More about her books is available at https://www.amazon.com/author/margaretreece. You may contact Dr. Reece at DrReece@MedicalScienceNavigator.com, or on LinkedIn.
Reece Biomedical Consulting LLC Privacy Policy: Click HERE
Featured header image: ©Moonlight Photo Studio, via Shutterstock.com