Article #4 of 7 Part Series
Neurons, the principle electrical cells and unmistakable superstars of the brain cell community, occupy the central hub of almost all efforts to clarify how the human brain works. It is agreed in general that these remarkable cells form the physical substance of the mind and a person’s sense of self.
Yet, neuroscientists agree on only some of the details in theories of how neurons communicate with each other. Few undisputed facts support the many ideas about how neurons create something as insubstantial as thoughts. One way to evaluate current knowledge of how the human brain works is to consider it a fuzzy snapshot with too few pixels.
This article is confined to a discussion of what scientists do know about how neurons send signals to each other and about how neurons are dependent upon the other 90% of cells in the brain for their optimal function. Several brain processes are highlighted that scientists think may be where the brain’s performance goes awry with injury or disease.
Neuron Physiology
Deciding where to begin when describing how neuron signaling occurs through neuron synapses is a challenge, because it can be a circular narrative. The receiving parts of one neuron respond to input from the messaging end of other neurons.
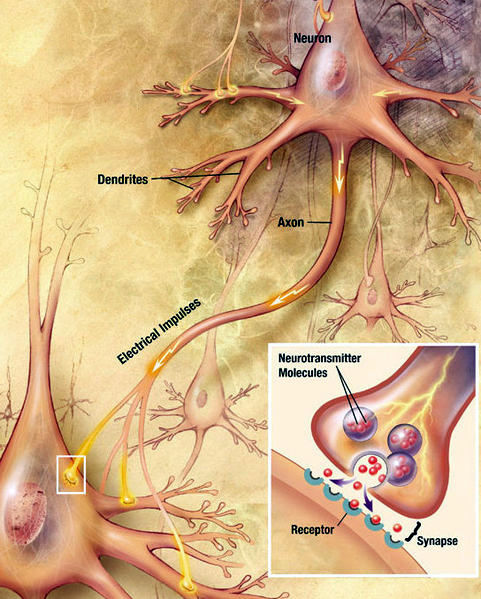
Neuron signaling through chemical synapses, National Institutes of Health United States Public Domain Illustration/Wikimedia Commons
The recipient neuron, in turn, sends a message about the net signal it receives as output to the next neuron. It is a net output signal, because input from connecting neurons may be positive or negative in nature.
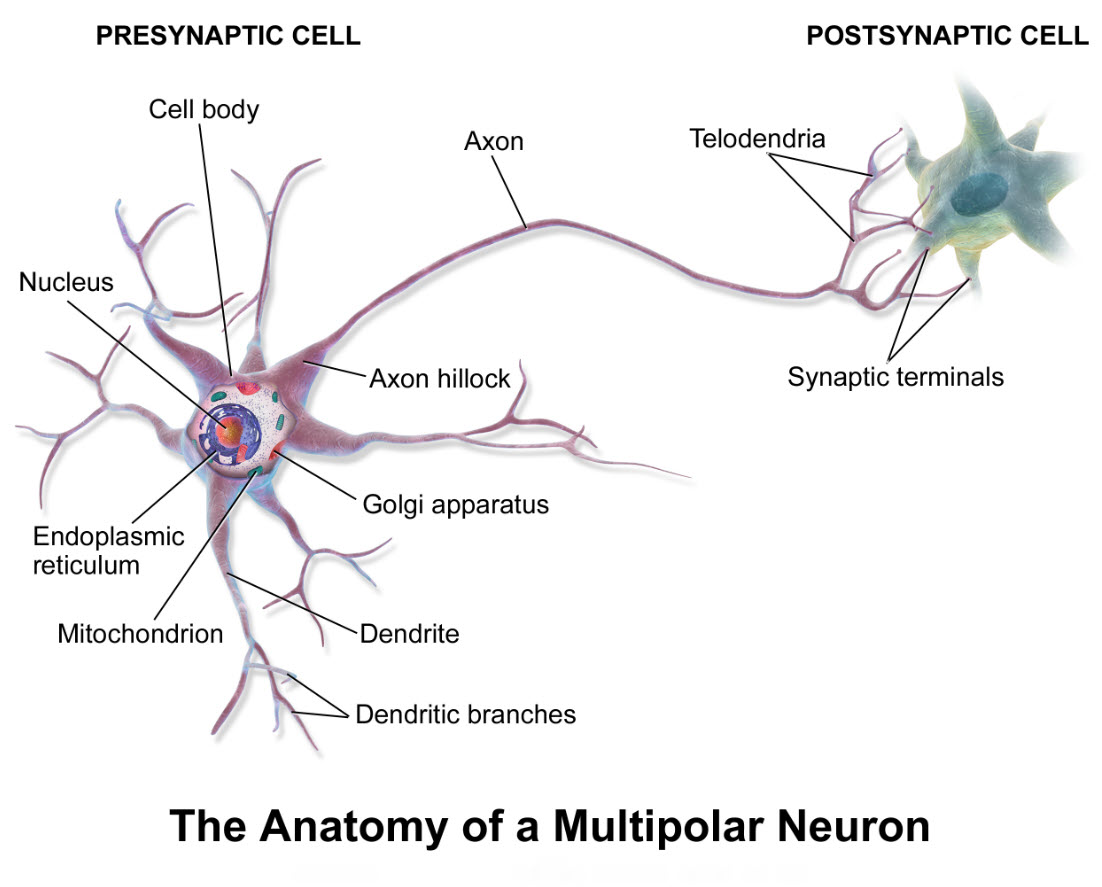
The narrative is further complicated by the fact that a neuron may receive signals from other neurons on its cell body, its dendrites, its dendritic spines, its axon and/or its axon terminal.
A neuron may even have a branched axon with one extension feeding back on itself. All these possibilities create an intricate network of neuron linkage called circuits.
The insert in the lower right corner of the illustration is a magnification of the most common type of brain axon-terminal connection, a synapse. Notice the synapse shows the neurons as separate cells communicating with each other by release of chemical molecules, neurotransmitter.
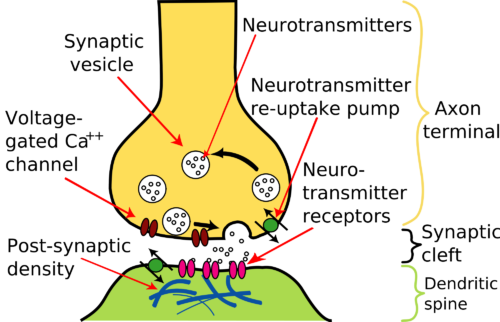
Neurotransmitter is stored in the axon terminals in vesicles and is released only when an electrical impulse reaches the terminal.
The section of axon terminal dedicated to neurotransmitter release is called the presynaptic compartment. Neurotransmitter released from the presynaptic compartment diffuses into a gap of about 20 nanometers between the presynaptic compartment and a modified section of the neuron cell membrane across the gap named a postsynaptic density. The gap itself is called the synaptic cleft.
The result of neurotransmitter activity in synaptic clefts of the brain is a multifaceted process that is only partially understood. Disturbances of normal activity at synapses can lead to a wide variety of brain disorders, among them anxiety and depression.
Neuron Electricity
Neuron electricity is usually described as if the neuron’s axon exists without the rest of the cell. Life scientists like to divide a living system into compartments for study. It is much easier to isolate one small part of the whole and study it as if it were separate from the rest. Scientists then take what they discover and attempt to apply the newly learned principles to another compartment.
Knowledge of neuron electricity evolved from studies of fluctuations in the amount of charged chemical particles in the fluid near an axon membrane. Once the mechanism for creating electrical impulses along an axon was known, it was easier to investigate possible variations in the theme at the neuron cell body and dendrites.
Fluctuation of the electrical charge around an axon is created by a moving stream of atoms called ions. In the case of the neuron axon, the ions involved are sodium, Na+, and potassium, K+. In this week’s earlier discussion of fluid quality control in the brain, it was noted that there is precise regulation of the composition of the fluid bathing neurons. One result of brain extracellular fluid quality control is to keep the amount of K+ low, and Na+ high, opposite their level inside the neuron. This sets up a concentration gradient near the axon’s membrane. Under resting conditions, the inability of charged molecules to cross the membrane prevents diffusion of ions from where they are in high concentration to where their concentration is low.
However, regulated opening of channels in the axon membrane permits diffusion of K+ out of the axon and Na+ into the axon. This diffusing stream of ions creates the electrical impulses neurons use to communicate. Of course, the axon’s membrane channels for Na+ and K+ are only open part of the time. Their sequential opening and closing along an axon is a tightly controlled process.
In fact cell membrane channels are exclusive to one ion. Na+ does not fit through a K+ channel, and a K+ does not fit through a Na+ channel. This allows diffusion of Na+ and K+ through axon membrane to be managed separately such that electrical impulses progress in one direction, from neuron cell body to axon terminal.
Neuron Synapses
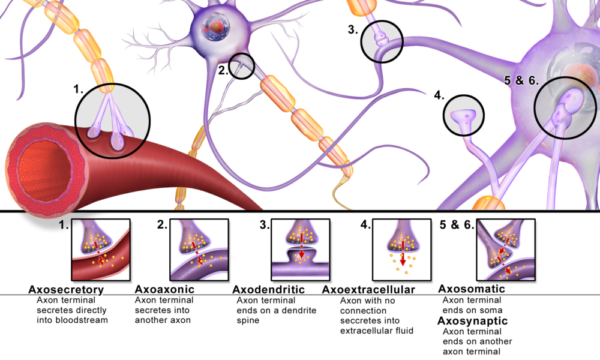
It is at neuron synapses that communication within the brain happens. A synapse is where a neuron’s axon terminal releases a chemical message called a neurotransmitter that diffuses across a small gap to another cell. All six varieties of synapses pictured here occur in the brain. Axodendritic synapses, shown in the illustration as type number 3, is the one most frequently discussed in textbooks.
In addition to what is pictured here, studies of the past 10 years discovered that there are two more cellular participants at each synapse.
Supporting the two neurons involved at a synapse is another star-shaped brain cell named an astrocyte. Astrocytes share metabolic pathways with neurons and remove neurotransmitter from synaptic clefts to terminate signals.
The fourth participant is another type of brain cell called microglia. Microglia monitors quality of the events at each synapse and repairs or removes postsynaptic components as needed.
Neurotransmitter Release
About a dozen different small molecules are used as neurotransmitter in the brain. Yet, the mechanism for neurotransmitter release from axon terminals is similar.
Neurotransmitter is made by metabolic enzymes in the neuron cell body and is packaged there into vesicles. The vesicles are transported along internal fibrous tracts through the axon to its terminal.
When electrical impulses arrive at the axon terminals, they cause calcium ion (Ca++) channels in the membrane to open. In adult brain the concentration of Ca++ is higher in the fluid surrounding axon terminals than inside the terminal. When Ca++ channels open, Ca++ enters.
The presence of a high level of Ca++ inside the terminal sets in motion a series of events that moves the vesicles from their storage area to the membrane near the synaptic cleft. The membranes of the vesicles then fuse with that of the axon terminal such that their neurotransmitter is released into the synaptic cleft.
The postsynaptic density is a thickening of the postsynaptic membrane that contains a large collection of proteins that respond to neurotransmitter called receptors. The diversity and size of the receptor population is governed by the chemical nature of neurotransmitter released and the pace of signaling from the axon terminal.
Some neurotransmitter receptors open ion channels. The ion channels may permit diffusion of Ca++, chloride ion (Cl–) or Na+ into the postsynaptic compartment, or K+ out of the compartment.
Other neurotransmitter receptors are proteins that penetrate through the postsynaptic density membrane. The trans-membrane receptors alter their shape when neurotransmitter binds. The modification of the receptor’s shape on the inside of the cell activates many molecular pathways leading to long-term changes in the postsynaptic density.
Brain Glial Cells
Only 10% of brain cells are neurons. The remaining 90% of cells participate as partners of the neurons in management of brain function. The general name for the non-neuron cells is brain glial cells. The name glia originates from a Greek word meaning glue. Early brain anatomists mistook the glia for a physical support system to hold the neurons together.
There is also a group of small cells in the brain once thought to be part of glia named microglia because of their small size. However, observations in recent years established microglia originates from a separate stem cell population than neurons and glia.
Astocytes
Astrocyte glia outnumber neurons by 5 times. They tile the brain in a non-overlapping arrangement. Astrocytes arise during development from the same stem cell population as neurons. It appears most of the original astrocyte population of fetal development survives most of a lifetime.
There are two types of astrocytes, protoplasmic and fibrous based upon their morphology and where they are in the brain. Protoplasmic astrocytes surround neuron cell bodies. Fibrous astrocytes locate near neuron axons.
Both types of astrocytes make extensive contact with blood vessels and regulate brain’s blood flow by secreting molecules to either dilate or constrict the vasculature. This effort by astrocytes insure local delivery of oxygen and glucose to meet the needs of highly active neurons.
Oligodendrocytes
Oligodendrocytes insulate neuron axons by wrapping themselves around them. Oligodendrocytes select larger axons, greater than 0.2 micrometers. A single oligodendrocyte often envelopes about 60 axons simultaneously.
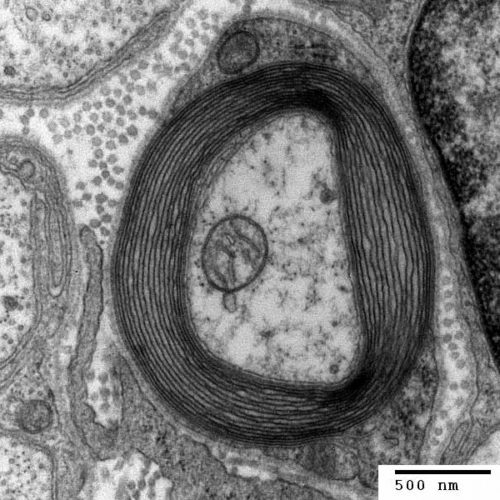
Cross section of a neuron axon with its myelin sheath, Generated and deposited into the public domain by the Electron Microscopy Facility at Trinity College/Wikimedia Commons
Upon contact with neuron axons, the exploratory membrane extensions of migrating oligodendrocytes convert to flat sheets that spread and wind around neuron axons to create a multilayered laminar cover. The laminar structure then compacts extruding almost all its intracellular material and becomes myelin.
Myelination of neurons in the human brain starts in late fetal life and the rate of myelination peaks in infants. Yet in certain regions of the cerebral cortex, neurons that control ongoing activity of life continue to increase their myelin into the 5th and 6th decades of life.
Microglia
Microglia, numbering approximately one per neuron, arranges itself throughout the brain in a non-overlapping grid. Each microglia cell monitor the environment around a single neuron.
Microglia originates from the extra-embryonic yolk sac during initial blood formation in early embryo genesis. Microglia have some of the same proteins as macrophages formed during blood formation by bone. However, microglia seeds the brain before blood formation begins in bone.
Microglia is present at all stages of brain development. In healthy brain, maintenance and expansion if the microglial population is dependent on self-renewal of resident cells.
Brain Function Partnerships
Astrocytes and Neurons
Astrocytes are necessary for neuron survival. Astrocytes form cellular webs, connected by gap junctions where the tips of their membrane extensions come in contact. Gap junctions, membrane structures with a central open channel, join the intracellular material of multiple astrocytes.
The larger astrocyte webs reach 1 millimeter in size and include hundreds to thousands of astrocytes. Astrocyte networks organize themselves into separate anatomic compartments. These astrocyte barriers may provide effective segregation of neuron activity in various parts of the brain.
In brain areas including the cerebral cortex and hippocampus, sites of memory formation and updating, the many branches of a single protoplasmic astrocyte may contact several hundred dendrites from multiple neurons and envelope up to 100,000 or more synapses. This affords individual astrocytes as well astrocyte networks extensive access to neuron circuits.
Most of the neurons that are active when the brain is awake release a neurotransmitter named glutamate. Glutamate is removed from synaptic clefts by astrocytes to quickly terminate each neuron signal.
About one third of the glutamate taken up by astrocytes is converted to another molecule called glutamine. Glutamine is returned to the neuron where it is converted back to glutamate. Astrocytes supplement glutamine made from synaptic neurotransmitter with glutamine synthesized de novo from glucose.
Neurons lack the enzymes necessary to make glutamate from glucose. One theory is that this arrangement provides an energy advantage to neurons. It permits ALL glucose entering neurons to be efficiently turned into high energy molecules rather than directing part of it to neurotransmitter synthesis.
Neurons require a great deal of energy. Neuron activity in the brain consumes about 20% of the body’s total energy production.
Oligodendrocytes & Neuron Axons
Oligodendrocyte insulate the axon of the neurons by wrapping them in their myelin. The layer of myelin along an axon is not continuous. Between myelin layers bare patches of axon occur. The bare patches are called Nodes of Ranvier. The Nodes of Ranvier contain the Na+ and K+ channels used to create the axon’s electric impulses.
Variation in myelin thickness and the length of the Nodes of Ranvier may provide opportunities for neurons to adjust the speed of their electrical impulses.
Neuron transport systems and overall neuron viability also appear to depend on supportive signals from oligodendrocytes. Axons with damaged myelin sheaths display alteration in the stability of their transport systems for carrying material from the cell body to the axon terminals.
Oligodendrocytes may also play a larger role in establishing electrical circuits in the brain than is appreciated. Axons often branch sending impulses from a single neuron to several locations for synapse.
Axon branching and axon/axonal synapses always occur at Nodes of Ranvier. The varying length of Nodes of Ranvier observed throughout the brain affect neurons’ ability to use branched axons and axon/axonal synapses in brain circuitry.
Microglia & Neurons
Microglia switches between two forms, surveying microglia and amoeboid microglia. The behavior of surveying microglia is regulated and guided by activity at individual neuron synapses. A high amount of activity in the synaptic cleft induces microglia to polarize its membrane extensions toward the synapse.
When neuron activity slows, surveying microglia removes structures from the dendrites named dendritic spines. It is at dendritic spines that most synapses occur. Pruning of synapses receiving little neurotransmitter input is required to maintain the mobile character of brain’s neuron circuitry.
Injured brain tissue releases factors that draws surveying microglia to the site. At the site of injury, microglia makes an executive decision whether neurons may be salvaged or not.
For salvageable neurons, microglia releases factors needed for their repair, and it recruits oligodendrocyte stem cells for replacement of axon myelin.
If damaged neurons cannot be repaired, microglia assumes its amoeboid, macrophage-like form and releases lethal compounds to finish the kill. It then it removes the debris by engulfing it.
We have covered a great deal today. But, if you would like further information about axon electrical impulses or brain synapses you can find it at “Neurons: Where does their electricity come from?” and “Brain Synapses” on this website.
 Margaret Thompson Reece PhD, physiologist, former Senior Scientist and Laboratory Director at academic medical centers in California, New York and Massachusetts is now Manager at Reece Biomedical Consulting LLC.
Margaret Thompson Reece PhD, physiologist, former Senior Scientist and Laboratory Director at academic medical centers in California, New York and Massachusetts is now Manager at Reece Biomedical Consulting LLC.
She taught physiology for over 30 years to undergraduate and graduate students, at two- and four-year colleges, in the classroom and in the research laboratory. Her books “Physiology: Custom-Designed Chemistry”, “Inside the Closed World of the Brain”, and her online course “30-Day Challenge: Craft Your Plan for Learning Physiology”, and “Busy Student’s Anatomy & Physiology Study Journal” are created for those planning a career in healthcare. More about her books is available at https://www.amazon.com/author/margaretreece. You may contact Dr. Reece at DrReece@MedicalScienceNavigator.com, or on LinkedIn
Reece Biomedical Consulting LLC Privacy Policy: Click HERE
Featured header image: ©Moonlight Photo Studio, via Shutterstock.com